April 6, 2026 by The Korea Advanced Institute of Science and Technology (KAIST)
Collected at: https://techxplore.com/news/2026-04-high-entropy-hydrogen-production.html
While mixing materials typically leads to instability, there exists a phenomenon known as high entropy, where increasing compositional complexity can actually enhance stability. KAIST researchers have leveraged this principle to enable faster proton transport and more efficient reactions within electrochemical cells, developing a technology that significantly improves hydrogen production efficiency. This breakthrough is expected to reduce hydrogen costs and accelerate the transition to clean energy.
New electrode tackles slow kinetics
A research team led by Professor Kang Taek Lee from the Department of Mechanical Engineering has developed a novel oxygen electrode material that dramatically improves reaction kinetics and power performance through entropy-maximized design. The oxygen electrode is a key component in electrochemical cells where oxygen evolution occurs during hydrogen production.
The paper is published in the journal Advanced Energy Materials.
Green hydrogen—produced from water without carbon emissions—is considered a cornerstone of future clean energy systems. In particular, protonic ceramic electrochemical cells (PCECs), which generate hydrogen by splitting water using electrical energy while protons migrate through the cell, have attracted attention for their high efficiency. However, their performance has been limited by slow reaction kinetics at the oxygen electrode.
High-entropy design of materials
To address this issue, the research team adopted a high-entropy strategy, introducing multiple metal elements simultaneously to increase configurational disorder. Although mixing many elements typically destabilizes structures, under certain compositions, maximizing entropy can instead stabilize a single-phase structure.
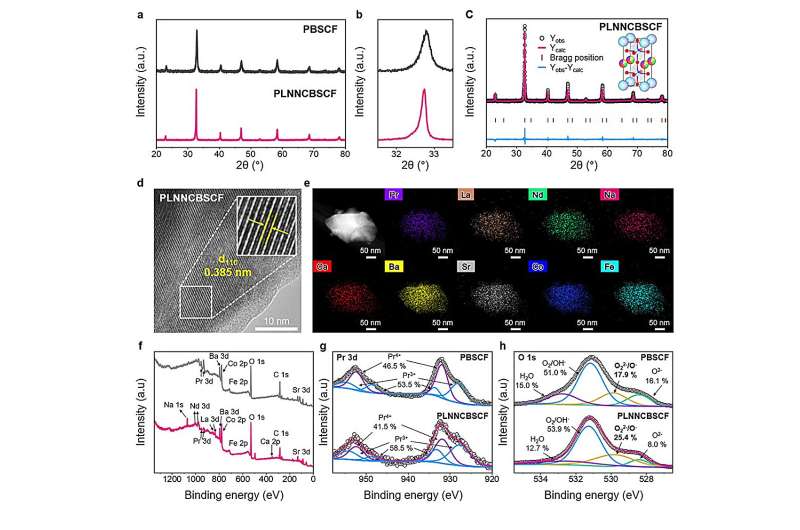
Based on this concept, the researchers designed a high-entropy double perovskite oxygen electrode by incorporating seven different metal elements (Pr, La, Na, Nd, Ca, Ba, Sr) into the A-site of the electrode structure. This material combines a perovskite crystal framework with a double perovskite structure, further enhanced by high-entropy design.
The presence of multiple mixed metal elements improves charge transport and oxygen-related reactions within the electrode, resulting in significantly faster electrochemical reactions for both electricity generation and hydrogen production.
Evidence from simulations and testing
Notably, density functional theory (DFT) calculations revealed that the energy required to form oxygen vacancies—active sites where reactions occur—was reduced by more than 60% compared to conventional materials. This indicates that reactive sites can form more easily and in greater abundance.
Additionally, time-of-flight secondary ion mass spectrometry (TOF-SIMS) analysis showed that proton transport speed increased by more than sevenfold, demonstrating that hydrogen generation processes proceed much more efficiently within the electrode.
The performance improvements were substantial. Cells incorporating the new electrode achieved a power density of 1.77 W cm⁻² at 650°C, approximately 2.6 times higher than conventional systems. Hydrogen production performance also improved by approximately threefold (4.42 A cm⁻²) under the same conditions.
Moreover, in long-term testing under steam conditions for 500 hours, performance degradation was only 0.76%, confirming excellent durability and stability over extended operation.
Professor Kang Taek Lee stated, “This study demonstrates that the thermodynamic concept of entropy can be used to control electrode reactivity. It has the potential to significantly enhance green hydrogen production efficiency and accelerate the commercialization of the hydrogen economy.”
Publication details
Seeun Oh et al, Unveiling Entropy‐Driven Performance Enhancement in Double Perovskite Oxygen Electrodes for Protonic Ceramic Electrochemical Cells, Advanced Energy Materials (2025). DOI: 10.1002/aenm.202503176
Journal information: Advanced Energy Materials

Leave a Reply