April 13, 2026 by Ingrid Fadelli, Phys.org
Collected at: https://techxplore.com/news/2026-04-phase-vo-methane-propane-hydrogen.html
Converting methane, the primary component of natural gas, into higher alkanes and hydrogen, could be highly advantageous. Alkanes, such as propane and butane, are easier to transport than methane and are used in a wider range of industries. Hydrogen, on the other hand, is a promising clean fuel used to power electrochemical devices that can generate continuous power, known as fuel cells.
Over the past decades, some energy engineers have been exploring the possibility of converting methane into hydrogen or complex hydrocarbons using photocatalysts. These are materials activated by sunlight or other types of light and that can drive chemical reactions.
Researchers at Université de Lille—CNRS, Sorbonne Université and other institutes in France recently introduced a new strategy for the photocatalytic conversion of methane into propane, which is widely used for heating, cooking, and transportation.
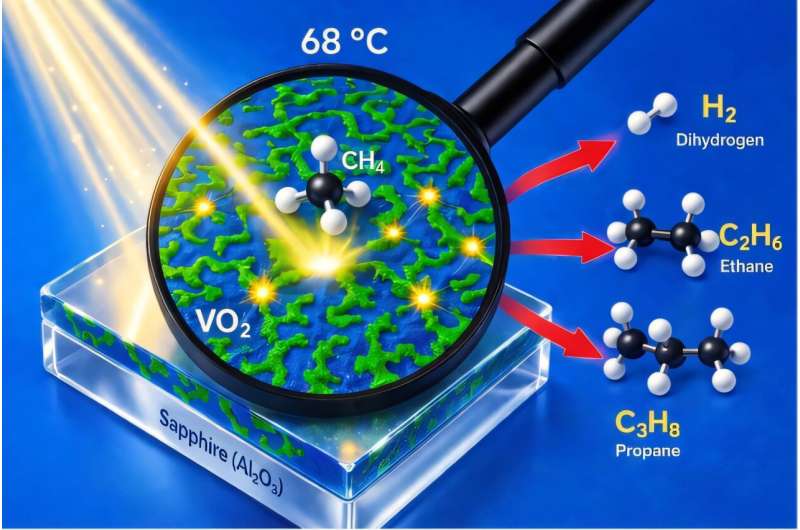
Their approach, outlined in a paper published in Nature Energy, leverages the properties of a material called vanadium dioxide (VO2), which changes from an insulating to metallic phase at a specific temperature.
“The main function of a photocatalyst is to activate chemical reactions to break down or transform molecules using the energy provided by light,” Bruno Grandidier, senior author of the paper, told Tech Xplore.
“A photocatalyst must therefore efficiently absorb photons to generate charges and ensure their efficient transfer to its surface. Unfortunately, for many materials used in photocatalysis, these charges recombine during their diffusion to the photocatalyst surface, thereby reducing its chemical power.”
To mitigate this widely reported limitation of photocatalysts, engineers often utilize electronic junctions. These are interfaces between different materials in a device or between different phases in a single material that can help to separate electrical charge.
“Inspired by recent developments in semiconductor photocatalysts using p–n or metal-semiconductor junctions, we were looking for a natural material in which junctions form spontaneously,” said Grandidier.
“We ultimately turned our attention to Mott insulators, materials characterized by a phase transition that causes them to switch from an insulating state to a metallic state under thermal, electrical or mechanical excitation.”
A new photocatalyst for methane conversion
VO2, the material used by the researchers, is not a pure Mott insulator, yet it exhibits Mott insulator-like behavior. Specifically, at a temperature of 68 °C, this material undergoes an electronic phase transition, during which co-existing metallic and insulating regions emerge.
“At the transition, VO2 contains two types of randomly distributed domains with contradictory properties,” explained Grandidier.
“In some places, the charges are delocalized and flow freely as in a metal, while in other places they are localized as in an insulator. The contact zone between the two types of domains forms a junction. It turns out that the distance between two neighboring junctions is much less than the diffusion length of the photogenerated charges, leaving them with enough energy to activate molecules on the surface of VO2.”
As part of their study, Grandidier and his colleagues fabricated VO2 films that varied in thickness, placing them on top of a sapphire substrate. They found that when the films were thin, they acted as reliable photocatalysts, enabling the efficient synthesis of hydrogen, ethane and propane from methane.
“Remarkably, a peak production occurred at a temperature of 68 °C, which corresponds to the temperature with the maximum of junctions in the film,” said Grandidier.
“A reduction of the VO2 film thickness caused a full selectivity for propane, making the exploitation of the metal-insulator transition in photocatalysis promising. Such a process could indeed offer an alternative to methane upgrading, an energy-intensive and CO2-emitting technique used by the petrochemical industry to meet the ever-growing demand for liquid propane.”
Towards the solar-powered synthesis of hydrogen and hydrocarbons
The approach for the photocatalytic conversion of methane devised by this research team so far proved to be particularly promising for the synthesis of propane. In the future, however, it could potentially be extended to the synthesis of hydrogen or of complex hydrocarbons beyond propane, such as butane and ethylene.
This study could also inform the design of other photocatalysts based on other phase-changing materials like VO2. Meanwhile, the researchers plan to continue refining their methane conversion strategy to further boost its energy-efficiency and selectivity (i.e., the amount of propane produced in proportion to other byproducts).
“The insulator-metal transition could also be triggered electrically at a lower temperature,” added Grandidier.
“Thanks to the applied electric field, which significantly reduced charge trapping by atomic defects in the film, record conversions of methane per gram of vanadium were reached. Investigating further the role of the electric field to make the reaction selective will be key to approaching the minimum thermodynamic energy needed to convert one mole of methane into propane, about 200 kJ per mol of methane.”
Publication details
My Nghe Tran et al, Exploiting the insulator–metal transition of VO2 in photocatalytic methane conversion, Nature Energy (2026). DOI: 10.1038/s41560-026-02013-w.
Journal information: Nature Energy

Leave a Reply