By Dalian Institute of Chemical Physics, Chinese Academy Sciences March 21, 2026
Collected at: https://scitechdaily.com/new-quantum-mechanism-boosts-energy-transfer-in-nanomaterials/
A newly identified mechanism reveals how proton motion can subtly yet powerfully influence triplet energy transfer in advanced materials.
In chemistry, some of the most important reactions do not depend on electrons alone. Protons can move at the same time, and that partnership helps drive processes that power life and modern technology.
Scientists already knew that proton-coupled electron transfer (PCET) plays a central role in cellular respiration, photosynthesis, nitrogen fixation, and energy-related materials. More recently, researchers added another piece to the picture with proton-coupled singlet energy transfer (PCEnT).
Building on these earlier discoveries, Prof. Kaifeng Wu’s team at the Dalian Institute of Chemical Physics, Chinese Academy of Sciences, set out to fill a key gap. They focused on how proton motion might be linked to triplet energy transfer, a process that differs fundamentally from singlet energy transfer but is equally important in both natural and synthetic systems.
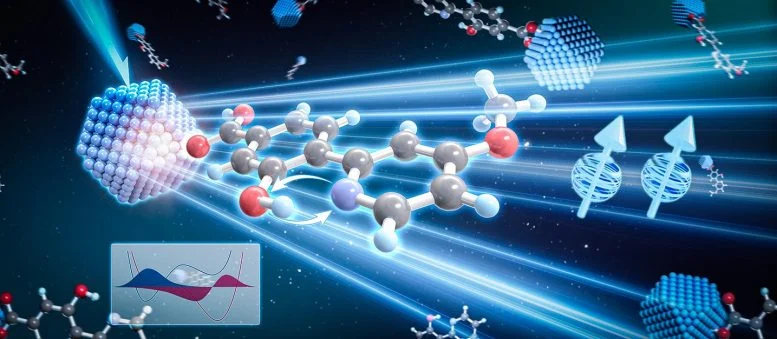
In a study published in Nature Materials, the researchers introduced a newly identified mechanism known as proton shuttle-assisted triplet energy transfer (PS-TET). This process takes place between ZnSe-based colloidal quantum dots (QDs) and phenol-pyridine molecular pairs attached to their surface.
Mechanism of Proton Shuttle-Assisted Transfer
When ZnSe quantum dots absorb light, they become excited and trigger a sequence of linked steps. First, a positive charge, or hole, moves from ZnSe to phenol while a proton shifts from phenol to pyridine. Next, an electron transfers from ZnSe to the phenoxyl radical, accompanied by the proton moving back from pyridinium.
Together, these steps enable the transfer of spin-triplet energy from the quantum dots to the phenol-pyridine system.
Even though the proton ultimately returns to its starting position, it plays a critical role as a shuttle. This temporary movement boosts both the speed and efficiency of triplet energy transfer compared to a similar system that lacks this feature. The team also found that adding a strongly electron-withdrawing trifluoromethyl group to pyridine can change the order of these coupled transfer steps.
Quantum Effects and Temperature Independence
The researchers observed that the PS-TET process remains largely unaffected by temperature. This suggests that the proton moves through quantum mechanical tunneling rather than classical motion. Their calculations of proton vibrational wavefunction overlap integrals support this interpretation.
These integrals influence how excited states relax and help direct energy along efficient pathways. The findings show that quantum effects can be used to control how charge and energy move through complex materials, even at room temperature.
“The discovery of the PS-TET mechanism has profound implications for many modern molecular technologies involving the spin-triplet excited states of molecules,” Prof. Wu noted. For example, improving triplet formation can enhance photoredox and environmental catalysis. On the other hand, devices like solar cells and lasers often need to limit triplet formation to perform efficiently.
The study suggests that adding or removing a proton shuttle could provide a way to control triplet formation on demand, offering new possibilities for designing advanced materials.
Reference: “Proton shuttle-assisted triplet energy transfer” by Zhaolong Wang, Jingyi Zhu and Kaifeng Wu, 9 March 2026, Nature Materials.
DOI: 10.1038/s41563-026-02535-4

Leave a Reply