March 17, 2026 by Apeksha Srivastava, Indian Institute of Technology Gandhinagar
Collected at: https://phys.org/news/2026-03-cow-dung-sustainable-carbon-dioxide.html
Climate change is one of the most pressing global challenges in the present times. Increasing carbon dioxide (CO2) concentrations in the atmosphere are a major factor contributing to this phenomenon. Activities such as the burning of fossil fuels for daily use, like electricity and transportation, and industrial applications, release significant amounts of CO2, trapping the heat at excessive levels and contributing to global warming.
How carbon capture technologies work
Carbon capture, use, and storage (CCUS) is one of the programs aimed at tackling fossil fuel emissions. The program explores various ways to capture CO2 so that it does not enter the atmosphere. Carbon capture can be accomplished through processes such as liquid chemical absorption, membrane separation, and adsorption using solid materials. While absorption involves soaking a substance into the entire volume of another substance, adsorption is the sticking of one substance to the surface of another.
Industries have been using these methods to let certain gases pass through while blocking or filtering out others. However, such processes face challenges, like high energy consumption, high development costs, and complex preparation processes, among others. A promising strategy is the use of porous carbons as CO2 adsorbents. They contain tiny pores that can trap CO2 on their surfaces. A good analogy is the way a charcoal air purifier removes invisible pollutants. But, the development of these adsorbents involves the use of corrosive and toxic materials, generates wastewater, and requires careful management to prevent environmental contaminants.
Cow dung as a clean resource
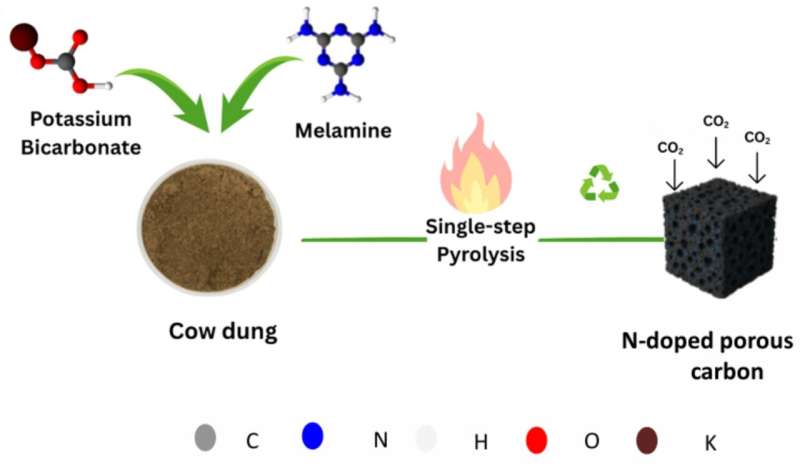
As an effort to address this issue, researchers from the Indian Institute of Technology Gandhinagar (IITGN) have proposed low-cost, sustainable, and scalable porous carbons derived from cow dung. Their findings were recently published in the journal Surfaces and Interfaces.
Cow dung is renewable in nature and available in large quantities daily in countries like India, where cattle are widely raised. In many rural areas, cow dung is commonly used as fertilizer and cooking fuel. In this study, it emerges as a high-value biomass precursor for producing advanced functional materials aimed at environmental remediation.
Engineering the nitrogen-doped porous carbon
Discussing the study, Mr. Ranjeet Kumar said, “We engineered nitrogen-doped porous carbon (NDPC), a material that adsorbs CO2. To make it, we mixed cow dung with melamine (which provides nitrogen) and potassium bicarbonate. The latter is a green, less corrosive, and effective activating agent to create high-surface-area materials.”
Mr. Kumar is the lead author of this study and a Ph.D. student in the Department of Chemical Engineering at IITGN.
“The mixture was heated in a furnace at an extreme temperature in the absence of oxygen. This process, called pyrolysis, produced NDPC for efficient capturing of CO2 molecules. Nitrogen incorporated into carbon improves this material’s ability to attract CO2,” added Sree Harsha Bharadwaj H, a co-author and a Ph.D. student in the Department of Materials Engineering at IITGN.
He employed detailed molecular-level simulations to validate the experimentally developed material. These methods simulate how atoms and molecules move over time and explore their possible configurations and properties.
Record surface area and performance
The researchers prepared different materials by changing the ratio of cow dung to melamine.
According to Dr. Chinmay Ghoroi, “The best-performing material, called NDPC-1, included 2.95% nitrogen and a very large surface area of 1,153 square meters per gram.”
Imagine one gram of the material covering several tennis court playing areas—that would provide some idea of how large this surface area is.
“The performance of NDPC-1 was 58% higher than that of pristine carbon, which is pyrolyzed cow dung without any activation. NDPC-1 also exhibited excellent cyclic stability, which is the ability of the porous carbon to maintain its initial CO2 capture capacity after 10 adsorption-desorption cycles,” continued Dr. Ghoroi, who is a professor in the Department of Chemical Engineering and the principal investigator at the ChemiGlyphic Lab.
Simulations reveal why it works
The team used an integrated approach to gain detailed insights.
In the words of Dr. Raghavan Ranganathan, “Instead of relying purely on experimental optimization, our work includes the combination of reactive molecular dynamics computational simulation and Grand Canonical Monte Carlo simulations with experiments to elucidate the structure-property relationship governing CO2 adsorption.
“This framework provides a deeper mechanistic understanding of nitrogen functionality and pore architecture, among other features. It also enables the development of high-performance porous carbons with optimized textural and chemical properties.”
Dr. Ranganathan is an associate professor in the Department of Materials Engineering and the principal investigator at the Computational Molecular Engineering Group.
Climate benefits and real-world promise
In essence, the NDPC-1 produced during this research has the potential to effectively capture CO2. Such materials, when combined with renewable energy technologies, will be crucial in reducing the carbon footprint of industries while making better use of resources. The material also supports the idea of a circular economy, which involves reusing waste materials to create valuable products.
Interestingly, the single-step and simple dry synthesis of the material reduces chemical usage and processing cost. Its decent adsorption capacity at low temperature and excellent regeneration stability make it a suitable and affordable candidate for large-scale deployment. The CO2 adsorption at low temperature (30° C) is a significant upgrade over most solid adsorbents that selectively adsorb CO2 at high temperatures (400–700° C).
Further, minimal wastewater generation and use of mild activating agents make the process environmentally sustainable. Importantly, this study aligns with the United Nations Sustainable Development Goal 13, centered on Climate Action, which encourages the world to transform its systems and deliver solutions to “preserve a livable planet.” The study is also in line with India’s National Action Plan on Climate Change, aiming for net-zero emissions by 2070.
More information
Ranjeet Kumar et al, Single-step dry synthesis of biomass-derived porous carbon for sustainable CO2 capture: Experimental and simulation study, Surfaces and Interfaces (2026). DOI: 10.1016/j.surfin.2026.108833

Leave a Reply